Why CTR-Test®?
Identifying Ineffective Chemotherapeutic Agents
- More than 95% accuracy for identifying ineffective chemotherapeutic agents before the start of a therapy.
- Applicable to any type of solid tumor (such as ovary, breast, lung, bowel).
- Taking specimens is possible from surgically removed tumor tissue, ascites or pleural effusion.
Advantages through Scientific Experiences
- Validated through 18 convincing clinical studies (on about 2,400 patients) and 11 more studies on tumor material from about 21,000 patients (see also Studies and Publications).
- CTR-Test® results correlate with response to therapy and the survival time of the patients.
Based on Trust
- About 150,000 patients have already used the CTR-Test®.
- More than 1,000 clinics worldwide have applied the CTR-Test® already.
- The CTR-Test® has been offered by TherapySelect since 2006 (see also Quality Management).
Humane
- Saves the patient from unnecessary toxicity.
- Valuable time for treatment is gained.
- Prevents cross resistance to otherwise effective therapies.
Cost Efficient
- Prevents costs caused by ineffective therapies.
- Spares costs accompanying those therapies (such as treatment of side effects).
What is the CTR-Test®?
The Cancer Therapy Response-Test (CTR-Test®) is a laboratory examination method, which is carried out before a chemotherapy. It is an in vitro diagnostic product capable of predicting resistance to chemotherapy of an individual patient with high accuracy (>95%) on the basis of living tumor cells, which have been removed from the patient. A double blind, randomized study has proven the reliability. Further studies show that survival also correlates with the test result in a statistically significant way and that the CTR-Test® is able to influence it.
High accuracy regarding the prediction of resistance is achieved because the CTR-Test® is a precise measure for tumor cell growth. The growth under the influence of various chemotherapeutic agents is determined over a longer period of time using drug concentrations, which can be found in patients.
In general, the decision in favor of a therapy is based on guidelines, phase-III-study results and the admission regulations for a specific chemotherapeutic agent. These depend on the type and location of the tumor, degree of metastasis, number of prior treatments and preferences of the individual patient (age, condition and side effects of prior therapies).
After taking these factors into account, the CTR-Test® is able to identify unsuitable therapies because it uncovers agents to which the tumor is very likely resistant. The decision in favor of a certain chemotherapeutic agent might therefore be influenced by the CTR-Test®.
It is possible to carry out the test on solid tumor tissue, ascites (malignant effusion inside the abdomen) and pleural effusion (malignant effusion inside the thorax). More detailed information can be found on the site Area of Application.More than
The drugs that are validated for the CTR-Test® can be found on the site Drug Selection.
By identifying ineffective substances, the test is able to spare patients unnecessary chemotherapies and the resulting side effects. Valuable time for different therapies is gained and the probability that resistance to other substances (so called crossresistance) arises is reduced.
Until now, 18 clinical studies with about 2,400 patients have been conducted on the CTR-Test®. All of them significantly verified the relevance of the CTR-Test® and benefit for the patients. 11 additional studies have validated the CTR-Test® on the basis of tumor material from about 21,000 patients.
The CTR-Test® has been carried out about 150,000 times. Therefore it is the leading technique to identify resistance to chemotherapy before administration to the patient and to spare the patient ineffective substances. Thus, the CTR-Test® definitely is no experimental method, but an established standard procedure. The CTR-Test® is the most established method for testing resistance to chemotherapy worldwide and is exclusively offered by TherapySelect.
If questions are unanswered to this or other topics yoMore thanu might find the answers on the site FAQ about CTR-Test®.
Process of the CTR-Test®
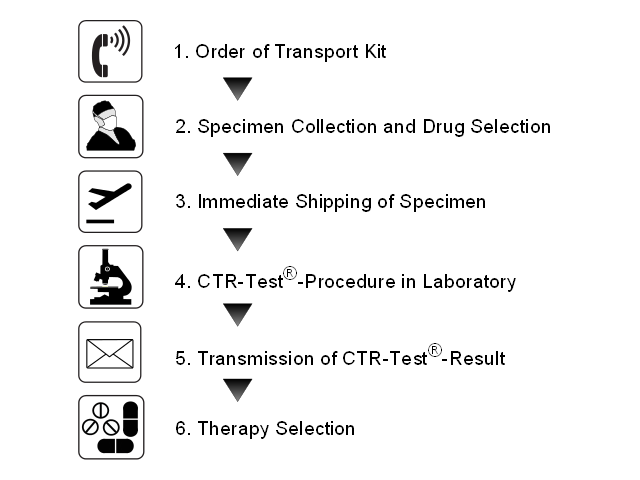
For the test viable tumor specimen of the patient is needed. For the transport a transport kit is provided. The kit should be present at the site before specimen collection. The treating physician selects the drugs to be tested. The tumor tissue is shipped to the laboratory of TherapySelect immediately after it has been removed from the patient. A highly qualified team of specialists processes the tumors and carries out the CTR-Test®. The result is sent to the treating physician 7 - 9 days after arrival of the tumor tissue at the laboratory. The oncologist is now able to select an optimized anticancer therapy for the patient.
Further information about the process and order of the CTR-Test® can be found on the site Process and Order and Documents for Download.
CTR-Test®-Procedure
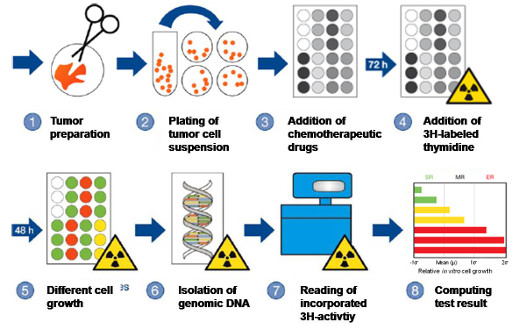
- Fresh, alive tumor tissue from the patient is cut up and digested enzymatically in order to obtain tumor cell structures (spheroids).
- The cells are kept under conditions, which favor growth of tumor cells and suppress growth of (possibly interfering) foreign cells.
- Subsequently, tumor cells are exposed to different chemotherapeutic agents for five days under carefully controlled growth conditions (treated cells are portrayed as colored dots, untreated cells are portrayed in white).
- Tritium-labeled thymidine is added during the last two days in order to enable determination of cell growth
- 48 hours later, tumor cells have grown differently strong (extreme cell growth, medium cell growth, slight cell growth)
- Newly formed, labeled genomic DNA is extracted
- The activity of labeled DNA is read by a scintillation counter and the cell growth is calculated using the data obtained.
- The growth of cells treated with chemotherapy is compared to the growth of the untreated control group (see white dots portrayed in step 3). If there are malignant cells present, which were exposed to a chemotherapeutic agent and grew as strong as cells from the untreated control group, administration of that agent to the patient will be ineffective with a probability of more than 95%. The length of the colored bars on the test result sheet indicates the level of resistance of tumor cells to the chemotherapeutic agents (Extreme Resistance, Medium Resistance, Slight Resistance).
CTR-Test®-Result
The decision in favor of a certain chemotherapy for treating a relapsing tumor disease is based on a variety of aspects. The first things to take into consideration are guidelines, phase-III-study results and admission regulations for specific chemotherapeutic agents. These depend on the type and location of the tumor, degree of metastasis, number of prior treatments and staging of the patients. Preferences of the individual patient (age, condition and side effects of prior therapies) are also included in the decision.
After taking these factors into account, the CTR-Test® is able to identify unsuitable therapies because it uncovers agents to which the tumor is probably (>95%) resistant. The decision in favor of a certain chemotherapeutic agent can therefore be influenced by the CTR-Test®.
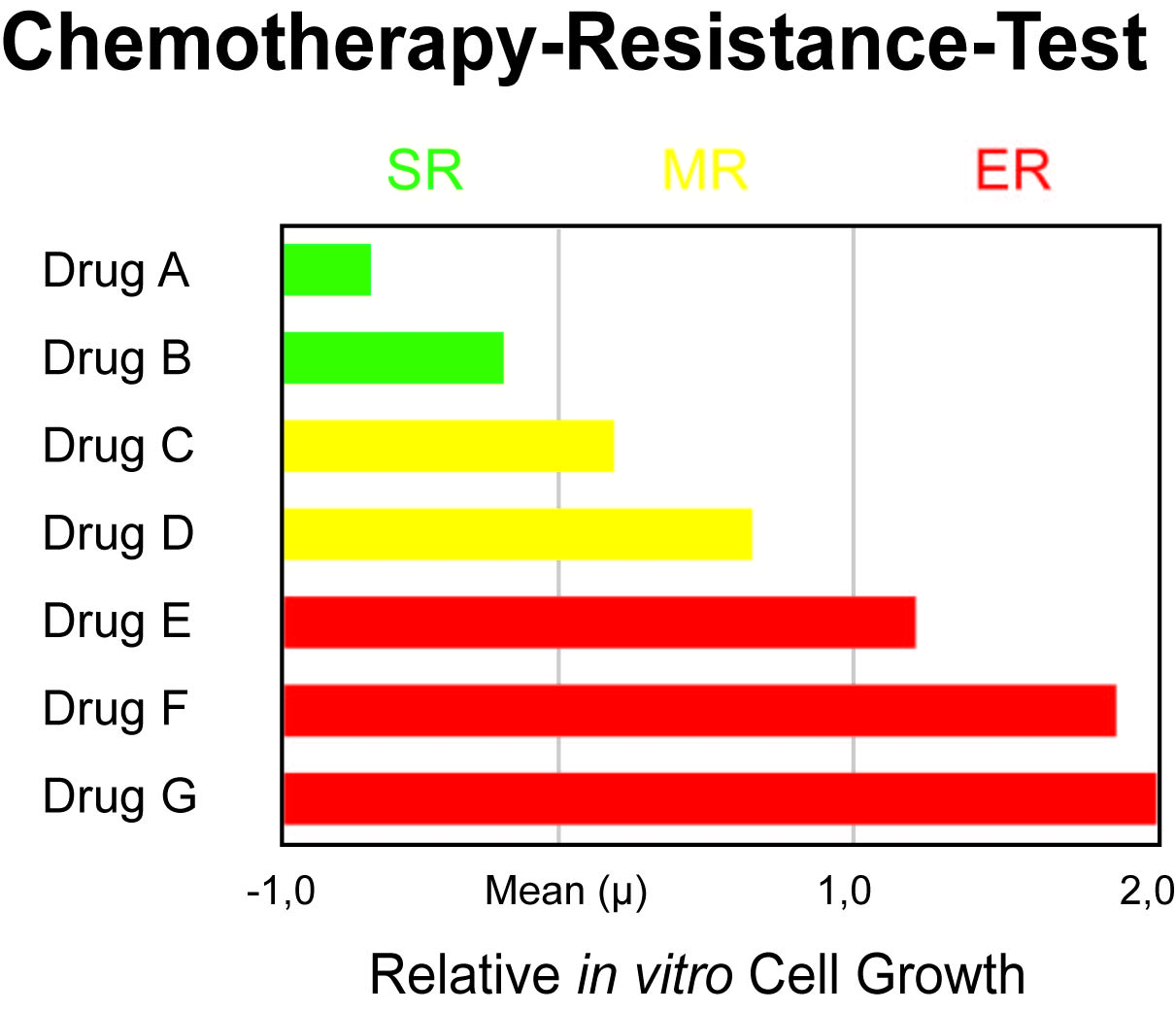
The following diagram shows an exemplary test result of a cancer patient. Seven agents were tested.
The tumor of the patient displays an Extreme Resistance (ER) to three chemotherapeutic agents. The physician is now able to exclude these ineffective drugs and to avoid unnecessary side effects. In this case, an alternative and effective therapy can be administered, which uses agents that were categorized as slightly resistant (SR). This increases the patient's chance to receive a successful therapy.
|
Clinical Evaluation of the CTR-Test®
The following information provides an overview of the clinical evaluation of the test. You will find a more detailed list of publications on the site Studies and Publications.
Correlation of the CTR-Test® Result and Clinical Response
The diagram shows the relation between the test result (in vitro resistance of a patient's tumor to a chemotherapy) and the actual clinical response of a patient to a chemotherapy. One dot represents one patient. Overall, correlations of 450 patients are depicted.
Patients were divided up into 3 groups (y axis) according to their test result:
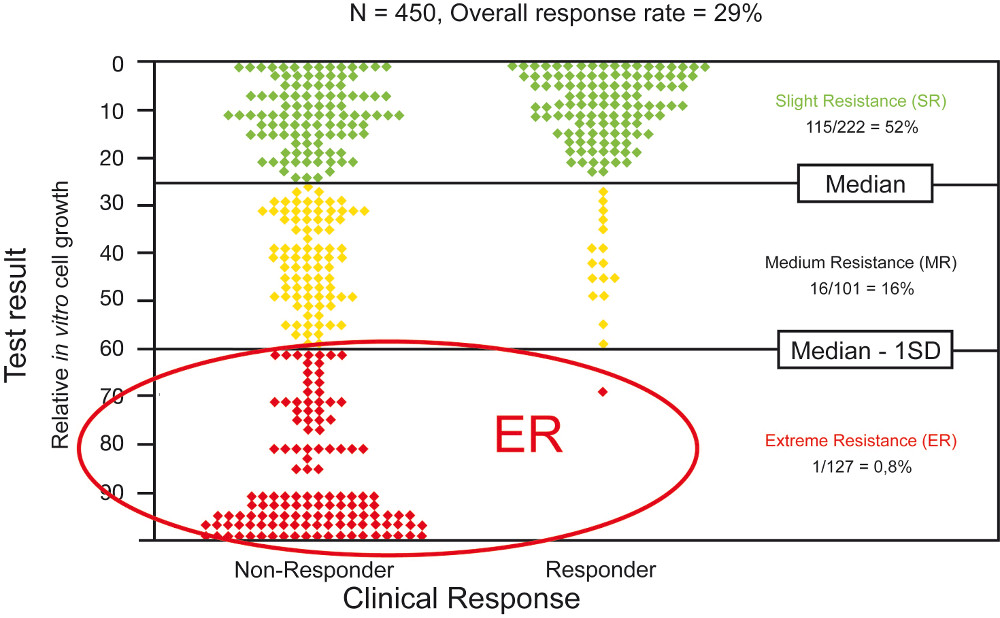
ER (Extreme Resistance): Tumor cell growth was more than one standard deviation from the median value. 126 patients (99.2%) out of 127 patients whose tumors showed an extreme level of resistance during the CTR-Test® did not respond to the chemotherapy.
MR (Medium Resistance): The level of tumor cell growth was higher than median growth, but smaller than one standard deviation above the median value.
SR (Slight Resistance): The level of tumor cell growth was below the median value. Tumor cells were only a little resistant to the used agent.
Correlation of CTR-Test® Results and Survival Time of the Patients
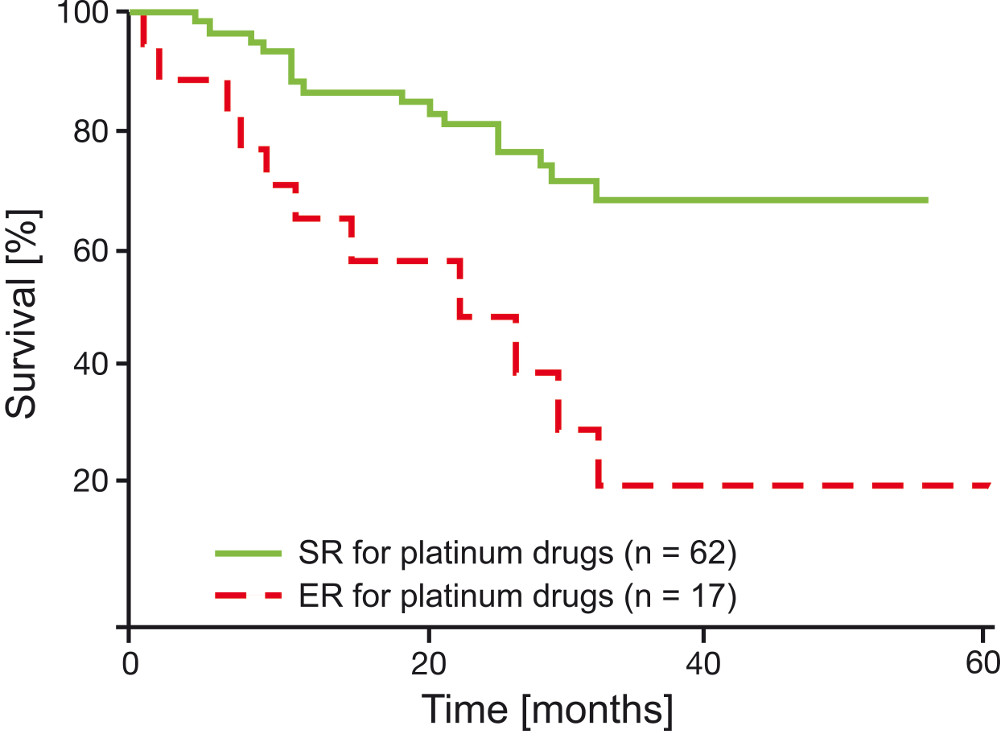
Holloway et al. (2002) demonstrated that the time until a relapse occurred and survival times were significantly shorter for patients whose ovarian tumors displayed resistance to platinum during the CTR-Test®, if they were treated with platinum based chemotherapeutics (p=0.0003).
Similar data was shown for breast cancer patients, who were administered combinational chemotherapy.
Extending Survival Time by Choosing a Chemotherapy Based on CTR-Test® Results
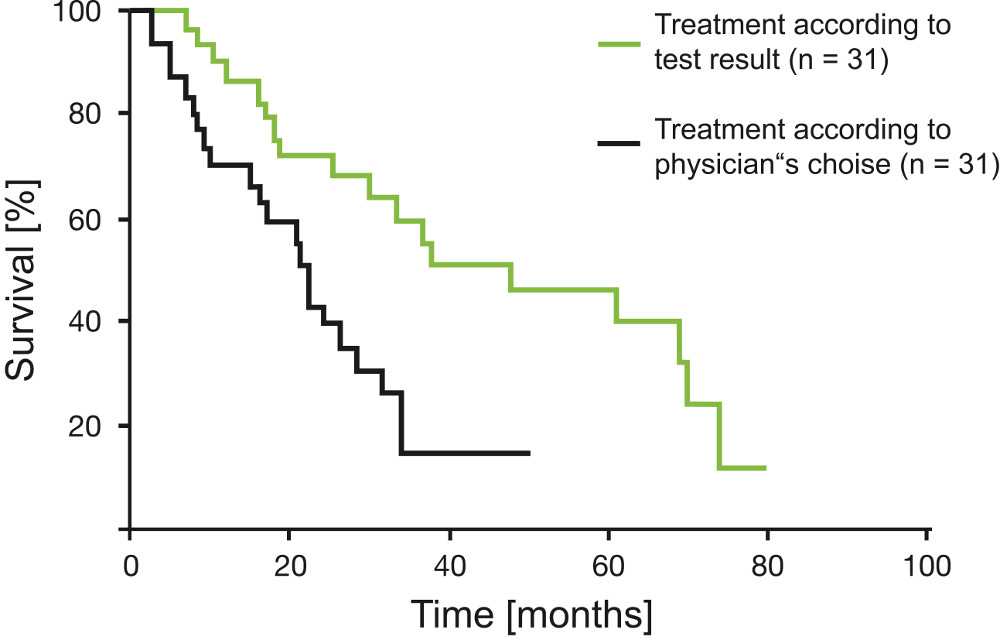
Loizzi et al. (2003) conducted a clinical study on platinum sensitive epithelial ovarian carcinoma. The outcome was that median survival time increased by 81% if ovarian cancer patients with recurrent tumors were treated with a therapy guided by the CTR-Test® result. Progression-free 1-year-survival rates were 90% in the group that was treated according to the test result compared to 70% in the control group (Treatment without test result) (p=0.005).
Prospective, Randomized Study on the Response of Primary Ovarian Carcinoma to a Platinum-Based Therapy
As part of the prospective, randomized, double blind EORTC study 55971 Verleye et al. (Oct. 2008) investigated the response of 246 primary ovarian carcinoma patients to a platinum based therapy. They showed that in vitro resistance to platinum based therapeutics as identified by the CTR-Test® correlates well with therapy failure in clinics. Therefore extreme resistance to carboplatin was an independent significant predictive factor for failure of response to first-line platinum-based treatment in advanced ovarian cancer.
Find out more:




