General Definition
Generally the term biomarkers comprises characteristic biological features, which can be molecular, anatomical, physiological or biochemical. These features can be measured and evaluated objectively, precisely and reproducibly. On the one hand they indicate the health condition of a patient and can for example provide information if a patient is already ill or how a disease will develop in future. On the other hand there are biomarkers which indicate the effect of a therapy or drug, respectively.
Examples for biomarkers providing information about the health condition are:
- the blood pressure level,
- the form of a mole,
- the amount of a protein or other substances in the blood or
- the alteration in the genome of cells.
Depending on the finding a clinical picture is concluded and the therapy is adjusted accordingly.
- A high blood pressure can be an indicator for an impending heart attack,
- an abnormally formed mole can indicate skin cancer,
- the presence of a high amount of sugar in the blood over a longer period can indicate diabetes and
- an alteration in the genome can indicate an increased risk for cancer.
Definition in Oncology
In oncology there are several types of biomarkers which provide information about the efficacy of cancer drugs.
Indirect efficacy testing for cancer drugs via molecular-based biomarkers
Molecular-based biomarkers are proteins and their function, DNA and RNA. The molecular-based biomarkers can be analyzed with the help of efficacy tests for cancer drugs. Thereby the presence (expression) of a protein or the presence of an alteration of the DNA (mutation) is examined for example. These molecular-based biomarkers show a predictive association with clinical response in clinical literature. This allows to draw a conclusion for the efficacy of drugs on cancer cells. For the efficacy prediction of a drug there can be one or more molecular-based biomarker.
A comprehensive consideration of molecular-based biomarkers is possible with the PCDx™ (Personalized Cancer Diagnostic).
Mutations and the associated efficacy of drugs can also be tested with a liquid biopsy with Guardant360®.
The molecular-based biomarkers normally deliver big data volumes and there is the challenge to identify the right biomarkers, which show clinical relevance. The above mentioned diagnostic methods are specialized for providing relevant information of this data volume for the physician and patient.
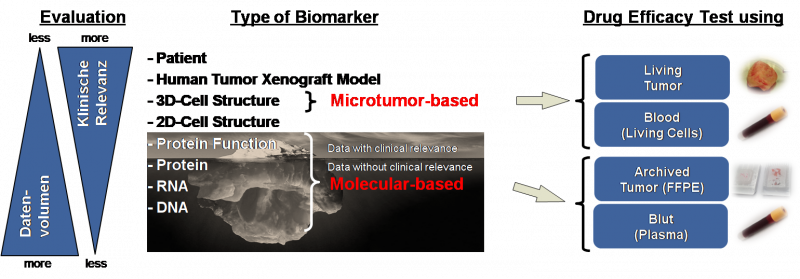
Direct efficacy testing for cancer drugs via microtumor-based biomarkers
Microtumor-based biomarkers represent an alternative for molecular-based biomarkers. These biomarkers use 3-dimensional cell structures (spheroids). Microtumor-based biomarkers normally show a higher clinical relevance as molecular-based biomarkers. The reason for this is that microtumor-based biomarkers (as for example the efficacy testing of drugs on the microtumor) approximately represent the cancer within a patient in the laboratory. That means the relevance of the efficacy prediction of drugs is better the more the complexity of the analyzing system resembles the disease of a specific patient.
Furthermore microtumor-based biomarkers provide a smaller data volume since the 3-dimensional cell structures can directly be treated and evaluated. On the contrary data volumes for molecular-based biomarkers can be very high. One example is the sequencing of genes to detect alterations (for example mutations, inserts, deletions, translocations), which are later associated with specific drug effects. That means relevant information, which could be relevant for the efficacy of a drug, have to be extracted from the big data volume. This topic is currently denoted Big Data.
A microtumor-based efficacy test for cancer drugs using 3D cell structures is the CTR-Test®. This test is able to predict the efficacy of chemotherapeutics.
A system which could simulate the conditions in patients even better are tumor xenografts. Thereby mice are injected with tumor cells of a patient. Afterwards the mice are treated with cancer drugs and it is investigated if the applied drugs have an effect on the tumor cells of the patient. However this is not applicable to clinical practice in order to determine the efficacy of cancer drugs for individual cancer patients.
For this reason microtumor-based biomarkers are the best method for efficacy testing of cancer drugs with the highest clinical relevance.